One of our general approaches to asymmetric amine synthesis involves stereocontrolled addition of free radicals to carbon-nitrogen double bonds, leading directly to the chiral alpha-branched amine (Reviews: Top. Curr. Chem. 2014, 343, 1; Tetrahedron 2001, 57, 5461). This approach, shown retrosynthetically (in reverse) below, is inherently more efficient than commonly employed methods where C-N bonds, C-C bonds and stereocenters are made in separate steps, for example via alkene epoxidation and subsequent epoxide opening with a nitrogen nucleophile. A major challenge which accompanies this approach is stereochemical control; this is still an unsolved problem in many radical reactions.

We have developed several ways to control the stereochemistry of radical additions to C=N bonds so that they are useful for real-life applications in synthesis of biologically active natural products and drug candidates. Our early work in this area led to diastereoselective additions of tethered carbon-centered radicals (J. Org. Chem. 2004, 69, 863), with successful applications to aminosugar synthesis (Org. Lett. 2007). We are also developing ways to control stereochemistry through the use of chiral N-acylhydrazones (Eur. J. Org. Chem. 2005, 3157). This provides excellent facial differentiation with a wide variety of radical precursors, including primary and difunctional halides (J. Am. Chem. Soc. 2000, 122, 8329; J. Am. Chem. Soc. 2001, 123, 9922; J. Org. Chem. 2006, 71, 7016). Either enantiomeric amine can be obtained by changing the roles of R1 and R2 groups in the figure below, or by using the enantiomeric auxiliary. Application to ketone hydrazones establishes access to alpha,alpha-disubstituted amino acids (Org. Lett. 2008, 10, 2311), and a general approach to gamma-amino esters was also established (Org. Lett. 2009, 11, 1095). This reaction now has the broadest scope and greatest flexibility among intermolecular radical additions to C=N bonds, including difunctional precursors leading to interesting annulation reactions (J. Org. Chem. 2015, 80, 6432), and has recently been adapted to photoredox catalysis (Org. Lett. 2019, 21, 8290).

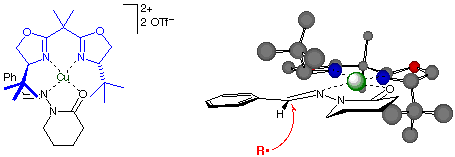
We achieved the first asymmetric catalysis of intermolecular radical addition to C=N bonds, using N-acylhydrazones as substrates and Cu(II)bisoxazoline complexes as chiral Lewis acids (Angew. Chem. Int. Ed. 2003, 42, 5061). High enantioselectivities (>90% ee) and good versatility for various hydrazones and various radicals make this a viable synthetic method at stoichiometric Cu loading. A tentative stereocontrol model consistent with the observed configurations is shown below. Catalyst turnover has been demonstrated, but high turnover numbers remain a challenging goal.