The inspiration for new methodology, as well as its proving ground, is total synthesis of natural products. We are targeting various natural products with biological activity of interest in antibiotic development, cancer research, neuroscience, and other areas of medicinal chemistry.
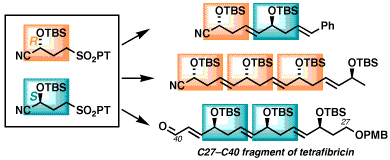
We are discovering interesting new methods to access alcohols and ethers in stereocontrolled fashion, and developing highly efficient strategies to take advantage of these reactions in complex molecule synthesis. Our targets possess important biological properties including antitumor, antibiotic, antimalarial and immunosuppressant activities. In 2010 we introduced a versatile strategy for rapid synthesis of 1,5-polyols (Org. Lett. 2010, 12, 5016), in which the configurations are encoded by asymmetric catalysis into enantiopure building blocks for iterative assembly. We have since expanded this to convert the 1,5-polyols into 1,5,7-triol motifs (Eur. J. Org. Chem. 2017, 1961). Two articles recently selected as Featured Articles in the Journal of Organic Chemistry describe the further development of this approach (J. Org. Chem. 2018, 83, 13636) and its application to synthesis and coupling of two fragments of tetrafibricin ( J. Org. Chem. 2018, 83, 13650 ) that were coupled using a Mukaiyama aldol reaction that revealed new insights on beta-silyloxy and beta-alkoxy groups on 1,3-diastereocontrol (Tetrahedron Lett. 2022, 111, 154203). We have also recently published a new route to alpha-silyloxyaldehydes, important building blocks for oxygen-rich natural products, using a novel three-step sequence from alkynes via ring-opening of enol ester epoxides (Tetrahedron Lett. 2012, 53, 5064).
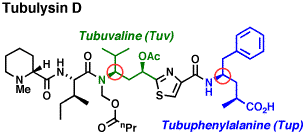
Total Synthesis of Tubulysins. Tubulysins are extraordinarily potent antimitotic agents currently in development for cancer chemotherapy. Two gamma-amino acid building blocks are the key structural novelty within the tubulysins. Using a C-C bond construction approach to the chiral amine moiety, we have developed new and efficient stereoselective syntheses of gamma-amino acids (Org. Lett. 2009, 11, 1095), and applied them to tubuphenylalanine and tubuvaline (Org. Lett. 2004, 6, 3249). Recent developments led to a highly stereoselective synthesis of a tubulysin tetrapeptide (J. Antibiot. 2016, 69, 294) and a second generation synthesis of tubuvaline (J. Org. Chem. 2021, 86, 15139).
Total Synthesis of Quinine. A longstanding challenge to molecular synthesis, the historic antimalarial agent quinine remains an intriguing target. Our interest stems from the potential to demonstrate new strategies for chiral amine synthesis, including a hybrid radical-ionic annulation. This key step of the synthesis combines a manganese-mediated radical addition to a chiral hydrazone followed by a nucleophilic cyclization to construct a substituted piperidine ring (Org. Lett. 2007, 9, 4243). This was followed by an oxidative ring cleavage and reclosure to form the azabicyclic ring system characteristic of the Cinchona alkaloids, completing a formal synthesis of quinine (Org. Biomol. Chem. 2011, 4039). Interesting computational results enlightened our understanding of the key ring closure in this synthetic route (J. Org. Chem. 2012, 77, 3159).